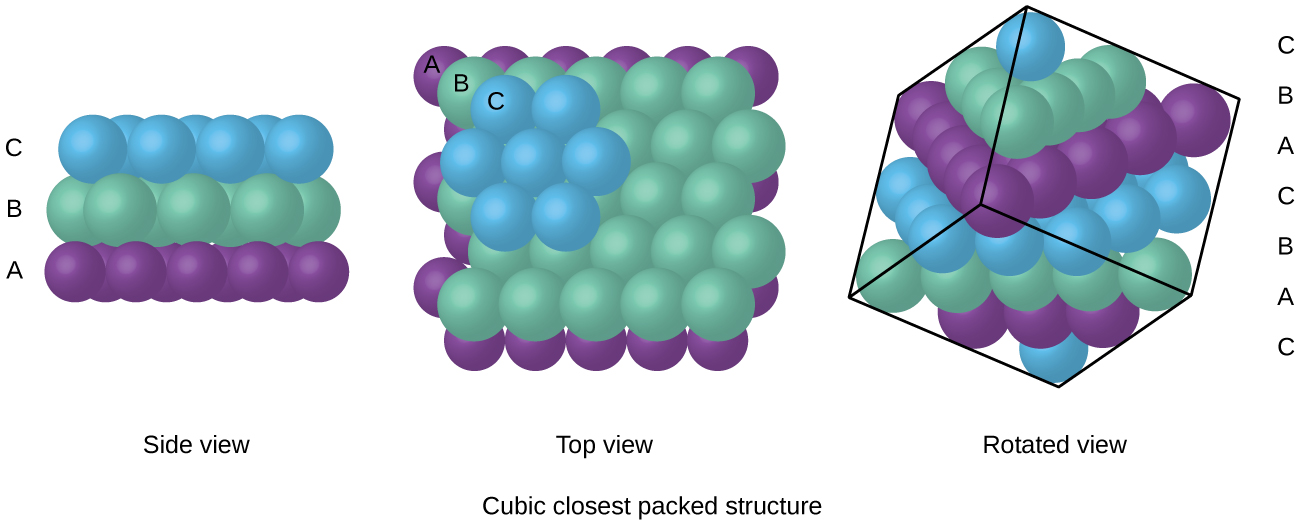
Stacking sequence is the order of the permanently repeating atom layers! Figure: Hexagonal closest packed (hcp) lattice structure (unit cell) This lattice plane sequence AB (also called stacking sequence) is now repeated continuously (ABAB …). An atom of the upper lattice plane thus sits in the gap formed by three atoms of the lower layer. However, it is just so shifted that the atoms of this plane fit exactly in the indentation of the underlying plane. The overlying atomic plane B (shown in blue) is constructed identically in principle. A single atom in this lattice plane A (shown in red) is always touched by a total of six neighboring atoms. In the hexagonal structure, the atoms of a lattice plane are densely packed together. The situation is different with the hexagonal lattice structure. The atomic levels are thus not maximally packed. In the body-centered cubic crystalline structure, the superposed atomic planes have gaps so that the respectively underlying plane fits exactly into the gaps of the upper one. The packing density (or packing factor) is the ratio of the atomic volume to the total volume of the unit cell! Figure: Derivation of the packing density of the body-centered cubic lattice structure (bcc) Hexagonal closest packed lattice structure The derivation of this packing density is shown in a separate post. The remaining 32 % are attributable to the “gaps” between the atoms. This means that 68 % of the total lattice volume (or unit cell) is occupied by atoms. For the body-centered cubic lattice, a packing density of 0.68 can be determined in this way. The packing density indicates what percentage of the unit cell is filled with atoms. If the atoms are realistically regarded as touching spheres, the so-called packing density of an atomic structure can be determined (also referred to as packing factor). Note, that these lines do not represent binding forces! Figure: Body-centered cubic unit cell (bcc) In order to clarify the spatial arrangement of the atoms, it is also common to indicate the basic form of the unit cell by dashes. The coordination number describes the number of directly adjacent atoms!įor the representation of unit cells, it makes sense to draw the atoms downsized, even if they “touch” in reality. In the simple cubic lattice, on the other hand, a coordination number of 6 is obtained. The coordination number in the bcc lattice is consequently 8. This number of direct neighbor atoms is also called coordination number. In the body-centered cubic crystal, one atom is in direct contact with eight surrounding atoms. Typical representatives of suchbody-centered cubic structures include the metals iron, chromium, molybdenum, vanadium and tungsten. The body-centered cubic lattice structure (bcc) contains another atom in the middle of the cube-shaped unit cell next to the corner atoms! Figure: Body-centered cubic (bcc) lattice structure (unit cell) In addition to the corner atoms, which each sit in the corners of the cube-shaped unit cell, there is now another atom centered in the middle of the cube (“body”). As with the primitive cubic lattice, the unit cell also has the basic shape of a cube. In this atomic structure, the individual planes of atoms (lattice plane) lie in the gaps of the respective lower planes of atoms. Body-centered cubic lattice structureĪ variation of the primitive cubic lattice is the so-called body-centered cubic lattice structure (bcc). For this reason, this article discusses the realization of the three most important lattice types. In order to be able to understand these differences in ductility, it is first necessary to understand the structure of the different types of lattice. Figure: Important lattice structures of metals The difference in the lattice structure has in particular strong effects on the ductility of the respective metals! While the fcc-lattice shows the highest ductility and the hcp-lattice the lowest deformability, the bcc-lattice has a malleability that lies between the other structures. These include the body-centered cubic lattice (bcc) and the face-centered cubic lattice (fcc), as well as the hexagonal closest packed lattice (hcp). Of much greater technical importance are variations of this simple cubic lattice structure. Figure: Simple cubic lattice structure (unit cell) Only in rare cases does a simple cubic crystalline structure appear, as shown in the animation below. The article on lattice structure of metals explains why the atoms in a metal are arranged with a certain regularity and thus form a lattice structure. 4 Face-centered cubic lattice structure.3 Hexagonal closest packed lattice structure.2 Body-centered cubic lattice structure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
